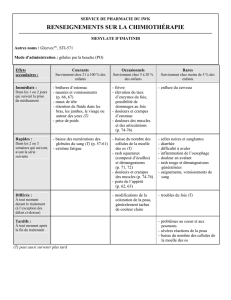
Pourquoi individualiser les doses de médicaments

Therapeutic Drug Monitoring
(TDM) of Tyrosine Kinase
Inhibitors (TKI) in MRCC
Réunion GSO Uro 19 Février 2014
1

TDM “Therapeutic Drug Monitoring”
•Determination of plasma concentrations as a
“routine” practice in order to adapt the individual
dose
•Prerequisite of TDM :
1) Prolonged treatment: daily oral dosage
2) Large interindividual PK variability, and limited PK intra-
individual PK variability
3) Low therapeutic index
4) Pharmacodynamic effects (efficacy and/or toxicity)
dose-dependent, and particularly concentration-
dependent
5) Possibility to define optimal concentrations 2

Inibs
•Imatinib: bcr-abl (Chronic Myeloid
Leukemia), cKit (Gastro Instestinal Stromal
Tumor)
•Nilotinib: bcr-abl (Chronic Myeloid
Leukemia)
•Erlotinib: EGFR (lung cancer)
• Sunitinib: VEGFR, cKit, … (renal cancer)
• Pazopanib: VEGFR, cKit, … (renal cancer)
3

2) PK variability
–elimination: clearance (CL)
–(distribution: volume of distribution (Vd))
–oral bioavailability (F)
4
Which PK parameters ?
Cmean,ss
=F(Dose/t)/CL
t

Elimination of inibs
•Liver metabolism (active or inactive metabolite)
–CYP3A4, CYP3A5 (pharmacogenetics)
–Drug-Drug Interactions (DDI) : e.g., CYP3A4 inducers
(carbamazepine, dexamethasone)
•Substrate of efflux transporters : ABCB1 (P-gp), ABCG2 (BCRP)
–Biliary and digestive elimination (parent compounds,
metabolites)
–DDI
–pharmacogenetics
•Elimination Clearance (CL)
–Ratio 10 between extreme values (CV50-70%)
–Liver impairment, elderly patients, children, …
–Non predictable in a specific patient 5
 6
6
 7
7
 8
8
 9
9
 10
10
 11
11
 12
12
 13
13
 14
14
 15
15
 16
16
 17
17
 18
18
 19
19
 20
20
 21
21
 22
22
 23
23
 24
24
 25
25
 26
26
 27
27
 28
28
 29
29
 30
30
1
/
30
100%