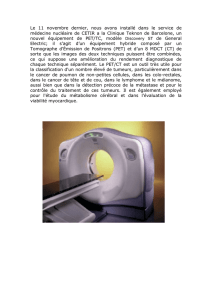
TE pancréatique fonctionnelle : insulinome


Pôle des Maladies de l’Appareil Digestif
Gastroentérologie-Pancréatologie,
Hôpital Beaujon, Université Paris VII, Clichy
NEUROENDOCRINE TUMORS
Diagnosis, etiopathogenesis and molecular biology
Philippe RUSZNIEWSKI
I GETNE EDUCATIONAL MEETING
Barcelona 7th April 2010

Beaujon Hospital group
for the study of NEuroendocrine Tumours
(NETs) of the digestive tract
French Referent Center for the study of NETs
Gastroenterology-Pancreatology:
Pr P. RUSZNIEWSKI (ENET Vice President), Pr P. HAMMEL, Dr O. HENTIC
Oncology:
Pr E. RAYMOND, Pr S. FAIVRE
Surgery:
Pr A SAUVANET, Dr S DOKMAK, Pr BELGHITI
Pathology:
Dr A. COUVELARD
Imaging:
Pr V. VILGRAIN, Dr M. ZAPPA
Genetic:
Dr D. VIDAUD

Where are NETs localized ?
Plöckinger, Neuroendocrinology 2004
Modlin, Cancer 2003
Stomach (10%), Pancreas (10%)
Small bowel (30%)
Rectum (20%),
Appendix (20%)
Colon (<5%)
GI tract: 75% Broncho-pulmonary: 25%

(Survey, Epidemiology and End Results (SEER), US population 1974–2005) 2.5–5.0/100,000 Inhabitants)
Per 100,000 Population
This increase is most likely due to improved detection such as
the availability of advanced radiological and endoscopic imaging.
• Data from the US SEER database indicate an approximate 5-fold increase
in the incidence of NETs between 1975 and 2004
0,0
0,2
0,4
0,6
0,8
1,0
1,2
1,4
1973
1974
1975
1976
1977
1978
1979
1980
1981
1982
1983
1984
1985
1986
1987
1988
1989
1990
1991
1992
1993
1994
1995
1996
1997
1998
1999
2000
2001
2002
2003
2004
Lung
Appendix
Stomach
Colon
Small Intestine
Rectum
Caecum
Pancreas
Yao JC, et al. J Clin Oncol 2008;26:3063–3072
NET Site
NET are rare tumours… but their incidence is increasing !
 6
6
 7
7
 8
8
 9
9
 10
10
 11
11
 12
12
 13
13
 14
14
 15
15
 16
16
 17
17
 18
18
 19
19
 20
20
 21
21
 22
22
 23
23
 24
24
 25
25
 26
26
 27
27
 28
28
 29
29
 30
30
 31
31
 32
32
 33
33
 34
34
 35
35
 36
36
 37
37
 38
38
 39
39
 40
40
 41
41
1
/
41
100%