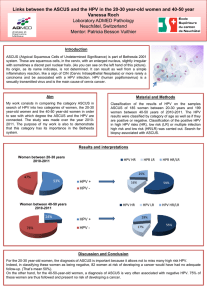
Current Scientific Initiatives Mechanisms of Carcinogenesis Section (MCA) Zdenko Herceg, PhD

Current Scientific Initiatives
Mechanisms of Carcinogenesis Section
(MCA)
Zdenko Herceg, PhD

Cancers are the consequence of combined genetic and
epigenetic changes induced by environmental
exposures

MCA Section
• The goal of the MCA Section is to advance understanding of
mechanisms of carcinogenesis and to contribute to biomarker
discovery
• This is achieved through investigating interactions between
the genes, the epigenome and the environment
• MCA also develops genetic/epigenetic methodologies that are
applicable to biobanks associated with epidemiological
studies
Research Groups
• Molecular Mechanisms & Biomarkers (MMB) – Magali Olivier (Acting)
• Epigenetics Group (EGE) – Zdenko Herceg

Early genetic changes (“drivers”) in cancer and their
links with environmental exposures
• Molecular events (somatic mutations) can be “read” as fingerprints of
exposures and mechanisms of mutagenesis
• Majority of studies investigated genetic changes in cancer tissues, less is
known about key early changes that promote tumour development
• Advances in genomics and major sequencing initiatives have opened an
opportunity for identification of target genes of environmental factors
Research aims
• Identify molecular events (mutations) that drive the process of tumour
initiation/progression and distinguish them from “passenger” events
• Identify environmental factors associated with these “driver” changes
• Contribute to the discovery and validation of new cancer biomarkers with
an emphasis on cancers associated with low-resource countries

Birth
Birth
Gametes
Gametes Childhood
Childhood Adulthood
Adulthood
Epigenome reprogramming
Epigenome reprogramming Cancer
Cancer
Leukemia
Leukemia
Parental exposures
Parental exposures Maternal diet/lifestyle
Maternal diet/lifestyle
ConceptionConception
In utero life
In utero life
Windows of susceptibility
Windows of susceptibility
Epigenetic profiling
Epigenetic profiling Cancer risk
Cancer risk
Exposure markers
Exposure markers
Impact of early life exposure on the epigenome and cancer risk
in childhood and adulthood
 6
6
 7
7
 8
8
 9
9
 10
10
 11
11
 12
12
 13
13
 14
14
 15
15
 16
16
 17
17
 18
18
 19
19
 20
20
 21
21
 22
22
 23
23
 24
24
 25
25
 26
26
 27
27
 28
28
 29
29
 30
30
 31
31
 32
32
 33
33
 34
34
 35
35
 36
36
 37
37
 38
38
 39
39
 40
40
 41
41
 42
42
 43
43
 44
44
 45
45
 46
46
 47
47
 48
48
 49
49
 50
50
 51
51
 52
52
 53
53
 54
54
 55
55
 56
56
 57
57
 58
58
 59
59
1
/
59
100%