Efficacy of Far Infrared Sericite Belt for Dysmenorrhea: Clinical Trial
Telechargé par
tartinet73

Complementary Therapies in Medicine (2011) 19, 187—193
available at www.sciencedirect.com
journal homepage: www.elsevierhealth.com/journals/ctim
A multicenter, randomized, double-blind,
placebo-controlled trial evaluating the efficacy and
safety of a far infrared-emitting sericite belt in
patients with primary dysmenorrhea
Chae Hyeong Leea, Ju-Won Roha, Chi-Yeon Limb, Jin Hwa Hong c,
Jae Kwan Leec, Eung Gi Min a,∗
aDepartment of Obstetrics and Gynecology, Dongguk University Ilsan Hospital, College of Medicine, Dongguk University, 814
Siksa-dong, Goyang, Gyeonggi-do 410-773, Republic of Korea
bDepartment of Medicine, Graduate School, Dongguk University, Seoul, Republic of Korea
cDepartment of Obstetrics and Gynecology, Korea University College of Medicine, Seoul, Republic of Korea
Available online 16 July 2011
KEYWORDS
Far infrared ray;
Primary
dysmenorrhea;
Randomized
controlled trial;
Sericite belt
Summary
Objective: To determine the efficacy and safety of a sericite which emits far infrared rays in
the management of primary dysmenorrhea.
Design: A multicenter randomized double-blind placebo-controlled trial.
Setting: One hundred four patients with primary dysmenorrhea were randomized to wear a
sericite or placebo belt during sleep for 3 menstrual cycles, and followed for 2 menstrual
cycles. Hot packs were used to heat the ceramics and ensure slight pain relief in both groups.
Main outcome measures: The main outcome measures were the severity of dysmenorrhea
assessed by a 10-point visual analog scale (VAS) and the number of patients who took analgesics
at each menstrual cycle. Safety was evaluated by physical examination and self-reporting of
adverse events.
Results: The baseline VAS scores were 7.27 ±0.19 in the experimental group and 7.38 ±0.19
in the control group. The severity of dysmenorrhea gradually decreased during the treat-
ment period in both groups, with major improvements observed in the third treatment cycle
(4.96 ±0.30 in the experimental group and 5.69 ±0.30 in the control group, p= 0.087). Dur-
ing the follow-up period, the decreased VAS score was maintained in the experimental group,
whereas the VAS score gradually returned to baseline in the control group, which resulted in sig-
nificant difference between the groups (5.08 ±0.31 vs. 6.47 ±0.31 at cycle 5, difference −1.39
[95% CI, −2.25 to −0.53], p= 0.0017). The number of patients who took analgesics decreased
in both groups, but the differences were not statistically significant. No serious adverse events
related to wearing the sericite belt occurred.
∗Corresponding author. Tel.: +82 31 961 7360; fax: +82 31 961 7155.
E-mail address: [email protected].kr (E.G. Min).
0965-2299/$ — see front matter © 2011 Elsevier Ltd. All rights reserved.
doi:10.1016/j.ctim.2011.06.004

188 C.H. Lee et al.
Conclusions: Our data suggest that a far infrared-emitting sericite belt with a hot pack might
be used as an effective and safe non-pharmacologic treatment option for women with primary
dysmenorrhea, with a prolonged effect after treatment.
© 2011 Elsevier Ltd. All rights reserved.
Introduction
Primary dysmenorrhea is defined as cramping pain in the
lower abdomen, occurring at the onset of menstruation,
in the absence of any identifiable pelvic disease. Men-
strual cramp is most severe during the first or second day
of menstruation and typically lasts for 8—72 h, and may
be accompanied by nausea, vomiting, fatigue, back pain,
headaches, dizziness, and diarrhea.1Dysmenorrhea is a
common disorder in women of reproductive age, with an
estimated prevalence of 18—81%.2About 5—14% of patients
with dysmenorrhea experience severe pain sufficient to
cause disturbances in daily activities and absenteeism from
work or school.3
Increased production of prostaglandins (PGs), especially
PGF2␣, by the endometrium and myometrium during men-
struation plays an important role in the pathogenesis of
dysmenorrhea.4,5 PGF2␣causes potent vasoconstriction of
the uterine blood vessels and myometrial contractions, both
of which reduce blood supply to the uterus.6The resul-
tant uterine muscle ischemia and hypoxia are thought to be
the origin of pain in patients with primary dysmenorrhea.7
Non-steroidal anti-inflammatory drugs (NSAIDs), such as
ibuprofen and naproxen sodium, and oral contraceptives
(OCs) are therefore the most common and rational choices
for the treatment of primary dysmenorrhea. NSAIDs suppress
endometrial PG biosynthesis by directly inhibiting cyclooxy-
genase activity. OCs reduce endometrial thickness, which
leads to a reduction in PG release.8However, 20—30% of the
patients with primary dysmenorrhea either do not respond
or obtain insufficient relief with these medications.7,9 More-
over, both treatments only have temporary effects and are
associated with some adverse events. NSAIDs are associated
with gastrointestinal upset or bleeding and serious kidney
or liver failure. OCs are associated with nausea, headache,
water retention, and vaginal bleeding and may cause venous
thromboembolism and cervical cancer with prolonged use.
Therefore, it is highly desirable to search for effective non-
pharmacologic alternatives that can be easily used for the
treatment of primary dysmenorrhea.
Sericite is a petrographic term used to indicate highly
refractive and fine-grained mica, usually muscovite in
composition, found in hydrothermally altered rocks.10 Tra-
ditionally, sericite has been used in alleviating pain in
the reproductive organs of Korean women.11 Since the
properties of far infrared (FIR) rays emitted from sericite
ceramics are known, they have been used in the medi-
cal field to promote health. We have numerous reports
that beds made from sericite ceramics help relieve dys-
menorrheic pain. FIR is an invisible electromagnetic wave
with a longer wavelength than visible light. FIR is part
of infrared radiation, which is subdivided into three cate-
gories according to different wavelengths, as follows: near
(0.8—1.5 m), middle (1.5—5.6 m), and far (5.6—1000 m)
infrared radiation.12 Thermal therapy using FIR has been
widely applied in medicine, including the treatment of
chronic fatigue syndrome,13 chronic pain,14 and wound
healing.12 FIR can penetrate human skin up to several cen-
timeters to exert its thermal effect.15,16 FIR might be useful
in reversing the reduced uterine blood flow through the
vasodilating effect in women with dysmenorrhea. Recently,
it has been reported in Korea that underwear emitting FIR
rays was effective in reducing the severity of dysmenorrhea.
The epidemiologic importance of primary dysmenorrhea
and the need for a safe, alternative strategy for the treat-
ment of dysmenorrhea prompted us to investigate the
efficacy of FIR in patients with primary dysmenorrhea. The
aim of the present study was to assess the efficacy of FIR-
emitting sericite belt in the improvement of dysmenorrhea
and in the reduction of analgesic use. Other outcome of
interest was safety evaluation.
Materials and methods
This multicenter, randomized, double-blind, placebo-
controlled study was conducted at 2 hospitals in Korea
between August 2008 and September 2009. The study was
conducted in accordance with Good Clinical Practice, guide-
lines of the International Conference on Harmonisation,
and the Declaration of Helsinki. The study protocol was
approved by an Institutional Review Board at each clini-
cal research center. All patients provided written informed
consent before enrollment.
Study population
Participants were recruited from the outpatient gyneco-
logical clinics in two university-based hospitals (Dongguk
University Ilsan Hospital and Korea University Guro Hospi-
tal) and through poster advertising placed in public areas of
the hospitals. Patients were first interviewed and screened
by gynecologists to determine eligibility for this study.
Patients filled out a questionnaire that asked about demo-
graphic characteristics, menstrual history, and medical and
reproductive histories, and underwent screening procedures
which included vital signs (blood pressure, pulse, respira-
tion, and temperature), laboratory tests (complete blood
count, hepatic and renal function tests, serum electrolytes,
urinalysis, urine hCG, and CA125), and gynecologic exam-
inations (Papanicolaou smear, bimanual examination, and
pelvic ultrasonography) to rule out secondary dysmenor-
rhea. Magnetic resonance imaging was performed if judged
clinically necessary. Patients were asked to place a mark on
the 10-cm line at a point that corresponded to the level of
pain intensity they felt. The visual analog scale (VAS) con-
sists of a 10-cm horizontal scale with the descriptors ‘‘no
pain at all’’ on the left and ‘‘the worst pain imaginable’’ on
the right. The distance in centimeters from the low end of
the VAS to the patient’s mark was used as a numerical index
for the severity of pain.

Efficacy and safety of sericite belt in women with primary dysmenorrhea 189
Patients meeting the following inclusion criteria were
eligible for enrollment: women between 15 and 45 years
of age; good health (determined based on medical history
and physical examination); regular menstrual cycles (28 ±7
days); pain score of ≥5 on a visual analog scale (VAS); and
required analgesic medication for at least 4 of 6 menstrual
cycles. Patients were excluded if they met any of the follow-
ing criteria: oral contraceptives use within 6 months before
the start of the study; wearing an intrauterine contraceptive
device; diagnosed with or suspected to have pelvic disease
such as endometriosis, adenomyosis, uterine myoma, ovar-
ian tumor, endometrial polyp, pelvic inflammatory disease,
and anomalies of the genital tract; pregnant or breast-
feeding; pelvic surgery ≥2 times; and incomplete menstrual
diary during the pre-treatment baseline period.
Study design
The study was conducted over 7 menstrual cycles as follows:
(A) pre-treatment baseline period for 2 menstrual cycles;
(B) treatment period (cycles 1—3); and (C) post-treatment
follow-up period (cycles 4 and 5). Before starting treat-
ment, all patients were observed for 2 menstrual cycles.
The participants were then randomly assigned into one
of the two groups (an experimental group treated with a
sericite belt and a control group given a placebo belt) at
the data coordinating center. The randomization sequence
was computer-generated using a block size of 6, as a 1:1 allo-
cation ratio to each treatment group with stratification for
the study center. The belts were labeled with the randomiza-
tion code and investigators, patients, and study statisticians
were blinded to the treatment allocation. Patients were
asked to wear the belt while sleeping at night for 3 consec-
utive menstrual cycles, after which patients were followed
up for 2 menstrual cycles without wearing the belt. Patients
were given a menstrual diary to record pain assessment using
the VAS beginning 2 days before the onset of menses to
the 4th day of menses, which was returned to the trained
study personnel within 7 days after each menstrual period.
Patients were allowed to take the medications that they
usually used for pain relief and instructed to record the
name and dose of the medication in the menstrual diary
throughout the study period. The use of other complemen-
tary therapies for dysmenorrhea was not permitted during
the study. Any adverse event was recorded, including the
severity, duration, and outcome.
Sericite belt and placebo belt
The sericite and placebo belts were manufactured as two
indistinguishable belts and designed to be comfortably
secured to the lower abdomen using an elastic band and
Velcro tape. The sericite produced at Gumcheon mines in
Korea was used in this study. Natural sericite powder was
baked at 1300 ◦C to create ceramics after several phases
of a purification process. The FIR emissivity and emission
energy of sericite ceramics were measured using Fourier
transform infrared spectrometer (Midac, Inc., Irvine, CA;
Model M2400 Series) at the Korea Conformity Laborato-
ries and the Korea Institute of Ceramic Engineering and
Technology. The sericite ceramics emit FIR with a peak wave-
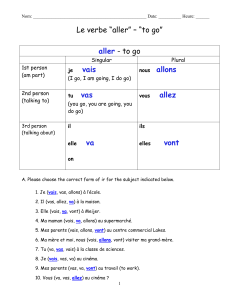
Figure 1 Elastic belt with button-shaped sericite ceramics
attached and a pocket behind them to keep a hot pack.
length of 5—20 m when warmed to a temperature of 40 ◦C.
As compared with black body, the value of emissivity of
sericite ceramics was 0.923 and the emission energy was
3.72 ×102W/m2. Forty-nine button-shaped sericite ceram-
ics were attached to the belt individually using strings
(Fig. 1). For the placebo belt, we used calcium carbonate,
so-called stone powder, instead of sericite. A pouch over the
ceramics in the belt was made to keep a hot pack. We used
9×7 cm sized disposable hot pack containing iron powder
and other chemicals. Hot pack produces heat through oxi-
dation process when it is exposed to air. It quickly heats up
to 50 ◦C and stays at that temperature for approximately
10 h. We included hot pack as a treatment in both groups
to heat the ceramics and to conform with the requirements
of the Ethics Committee to ensure slight pain relief during
treatment.
Outcome measures
The primary outcome parameter was the change in maximal
pain intensity using VAS during the treatment and follow-up
period. Among the VAS scores recorded in menstrual diary
from 2 days before the onset of menses to the 4th day of
menses, the highest VAS score was selected as the maximal
VAS of the menstrual cycle. The average of the two maxi-
mal VAS scores during the two baseline menstrual cycles was
used as the baseline VAS. Secondary outcome parameters
included the number of patients who took analgesics and
safety evaluation. Safety evaluation was based on physical
examination and occurrence of adverse events.
Statistical analyses
The intention-to-treat (ITT) population, defined as all ran-
domized patients who wore the sericite or placebo belt at
least once, was used in all efficacy and safety analyses.
Missing values were imputed using last-observation-carried-
forward (LOCF) method for subjects who did not complete
the study. The results of descriptive analyses at baseline
are reported as the mean and standard deviation. The dif-
ferences in baseline characteristics between groups were

190 C.H. Lee et al.
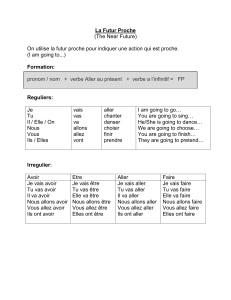
Assessed for eligibility (n=141)
Pre-treatment baseline period
(2 menstrual cycles)
Eldd( 37)
Randomized (n=104)
E
xc
l
u
d
e
d
(
n=
37)
-not meeting the inclusion criteria(n=7)
-menstrual diary not complete (n=30)
Sericite belt (n=52) Placebo belt (n=52)
Wear belt for 3 menstrual cycles Wear belt for 3 menstrual cycles
Lost to follow-op (n=6)
- Withdrawal of consent (n=6)
Lost to follow-op (n=9)
- Withdrawal of consent (n=8)
- No explanation (n=1)
Post-treatment follow-up period
(2 menstrual cycles)
Post-treatment follow-up period
(2 menstrual cycles)
Analyzed (n=52)
Analyzed (n=52)
Analyzed
(n=52)
Analyzed
(n=52)
Figure 2 Flow of patients through the trial.
examined using Student’s t-test or Wilcoxon’s rank-sum test
for continuous variables and Chi-square test for categorical
variables. The values of maximal VAS at each menstrual cycle
were compared between groups using analysis of covari-
ance (ANCOVA). The ANCOVA model included fixed effect
for center and baseline VAS value as a covariate. In addi-
tion to p-values, least square means for the two treatments
and differences between groups at each menstrual cycle
were estimated from the ANCOVA together with 95% con-
fidence intervals. For the primary endpoint, adjustments
for multiple comparisons with the use of a Bonferroni-Holm
procedure were incorporated into the analyses. The use of
analgesics and occurrence of adverse events between groups
were analyzed by Chi-square test and Fisher’s exact test,
respectively. All reported pvalues were from 2-sided tests
and considered significant at p< 0.05. All statistical analyses
were performed using SAS software version 9.2 (SAS Institute
Inc., Cary, NC, USA).
The sample size was estimated using the Power Analysis
and Sample Size for Windows software (Package PASS 2008,
NCSS, Kaysville, UT, USA). A sample size of 43 patients per
group was required to detect a mean VAS score difference of
2.0 between the experimental and the control group, with
at least 80% power and an ˛level of 0.05 (2-sided t-test),
Table 1 Demographic and menstrual characteristics of study population.
Sericite group (n= 52) Control group (n= 52) pvalue
Age 27.6 ±6.2 28.1 ±6.3 0.855
Height 162.1 ±5.0 162.2 ±5.0 0.794
Weight 55.2 ±7.3 55.5 ±7.4 0.883
Menarche (years) 13.6 ±1.4 13.3 ±1.3 0.140
Family history of dysmenorrhea 0.556
Yes 29 26
No 23 26
Parity 0.631
Nulliparous 42 40
Multiparous 10 12
Cycle length (days) 30.4 ±4.0 30.0 ±4.2 0.098
Maximal pain intensity (VAS) 7.3 ±1.4 7.4 ±1.4 0.663
Analgesic use during menstruation 0.497
Yes 36 42
No 16 10

Efficacy and safety of sericite belt in women with primary dysmenorrhea 191
Figure 3 Change in maximal VAS score between sericite and
control groups during the study period.
assuming a standard deviation of 2.73. We tried to recruit
51 patients per group into the study to compensate for an
expected dropout rate of 15%.
Results
Patient disposition and baseline characteristics
One hundred forty-one patients with dysmenorrhea were
recruited and screened for the study (Fig. 2). Thirty-seven
patients were excluded before randomization because they
were diagnosed with secondary dysmenorrhea (n= 7) or did
not fill out their menstrual diary completely during the
pre-treatment baseline period (n= 30). One hundred four
patients were randomized to wear a sericite (sericite group)
or placebo belt (control group), with all 52 women in each
group included in the analysis. Six patients in the sericite
group and nine patients in the control group were lost to
follow-up during the treatment period. Among 89 patients
who completed the study, 83 patients (41 in the sericite
group and 42 in the control group) wore the sericite or
placebo belt at least 70% of the total days of the 3 men-
strual cycles. The baseline characteristics are presented in
Table 1. There were no significant differences in age, age at
menarche, length of menstrual cycle, parity, maximal VAS
score, and analgesic use between the groups.
Efficacy
The change in maximal VAS score during the study period is
shown in Table 2 and Fig. 3. The VAS scores were highest dur-
ing the baseline cycle and were comparable between groups
(7.27 ±0.19 vs. 7.38 ±0.19, p= 0.664). The intensity of
menstrual pain decreased over time for both groups during
the treatment period, with major improvements observed
at the third menstrual cycle (4.96 ±0.30 vs. 5.69 ±0.30,
p= 0.087). Although both groups showed significant improve-
ments compared with the baseline VAS score, there were
no differences between the groups. During the follow-up
period, the earlier improvement in dysmenorrhea was main-
tained in the sericite group, whereas the improvement in
dysmenorrhea gradually returned to the baseline VAS score
in the control group. As a result, there was significant dif-
ference in the maximal VAS score at cycle 5 (5.08 ±0.31
vs. 6.47 ±0.31, difference −1.39 [95% CI, −2.25 to −0.53],
p= 0.0017) between the groups.
Thirty-six and 42 patients took pain medications at base-
line period in the sericite and control groups, respectively
(Table 3). Although the number of patients who used anal-
gesics decreased over the treatment period (29 in the
sericite group and 19 in the control group at cycle 3), no sig-
nificant differences were found between the groups. During
the follow-up period, the number of patients who took anal-
gesics further decreased from 29 to 26 in the sericite group,
whereas it increased from 19 to 25 in the control group.
However, there were no significant differences between the
groups.
Safety
Reporting of adverse events did not differ between the
groups (Table 4). Seventeen adverse events were reported
by 15 patients (14.4%). The most common adverse events
were first degree burns (41.2%) owing to the hot pack and
itching (35.3%), with an equal frequency in both groups.
All of these adverse events disappeared within a few days
without treatment. There were no serious adverse events
and no clinically relevant changes in vital signs. No patient
discontinued the clinical trial due to an adverse event.
Discussion
Our results indicate that, although not statistically signif-
icant between groups, the VAS scores decreased in both
groups compared with baseline during the treatment period.
However, during the follow-up period, statistically signif-
icant difference in the VAS scores between groups was
observed. This difference arose mainly because the reduced
VAS score was stable and maintained in the sericite group,
whereas it gradually increased in the control group. A sim-
ilar decrease in the VAS score during the treatment period
in both groups could be partly attributable to the effect
of the hot pack. Hot packs are known to have beneficial
effects in dilating blood vessels, increasing blood flow, and
influencing transmission of pain by reducing the level of
nociceptive stimulation.17,18 It has been demonstrated that
continuous low-level topical heat therapy with a patch or
a wearable heat wrap applied directly on the skin of the
lower abdomen provides significant pain relief in patients
with dysmenorrhea.19,20 However, the sustained pain relief
in the sericite group during the follow-up period suggests
that, although the effect was masked by the hot pack during
the treatment period, the sericite belt reduced the severity
of dysmenorrhea. In addition, there were no statistically sig-
nificant differences in adverse events, and the events were
generally minor and short in duration. This is the first clin-
ical trial to provide evidence that a sericite belt might be
an effective and safe non-pharmacologic alternative for the
treatment of primary dysmenorrhea.
Although the precise mechanism underlying sericite’s
efficacy for dysmenorrhea is still unclear, one possible
mechanism is the thermal effect of FIR rays from sericite,
 6
6
 7
7
1
/
7
100%