Social dominance and stress hormones

TRENDS in Ecology & Evolution
Vol.16 No.9 September 2001
http://tree.trends.com 0169-5347/01/$ – see front matter © 2001 Elsevier Science Ltd. All rights reserved. PII: S0169-5347(01)02227-3
491
Opinion
Animals respond to a stressor with a series of endocrine
responses that increases the immediate availability of
energy, in part by inhibiting physiological processes
that are not required for immediate survival1–3. One
of the primary responses to stress is an increase in the
activity of the hypothalamic–pituitary–adrenocortical
axis, causing an increase in the concentration of
circulating adrenal glucocorticoids (GC). In the short
term (hours to days), GC elevations redirect resources
to mobilize energy that can be used to resolve the
stressful situation2. If the stressor is not eliminated,
and GC levels remain high for more than a few days, a
broad range of harmful consequences ensue,
including immune suppression, loss of muscle mass
and reproductive suppression2,4,5.
Defining stress
Even among biologists, the word ‘stress’has many
meanings6. It can be used to refer to the condition
provoking a response (a stressor), and to refer to
changes in internal state induced by the external
condition (a stress response). It is generally clearer to
avoid the word stress, and refer explicitly to stressors
and stress responses. For the purposes of this review,
a stressor is any stimulus (behavioral, environmental,
or demographic) that provokes a physiological stress
response, as measured by an increase in GC secretion.
Although other physiological systems also respond
directly to stressors, GC responses have been studied
in a broad range of species, including some in the wild.
In addition, the physiological and behavioral effects of
GCs are relatively well characterized2,4 relative to other
stress responses, for example, the production of heat
shock proteins7. Even for GCs, there is considerable
debate about the physiological and behavioral
consequences of increased secretion3, and responses
to GC elevation vary among individuals and species8.
There is a tendency to consider any difficult or
energetically demanding situation a stressor. As
Wingfield and Ramenofsky6point out: ‘phrases such
as “the stress of reproduction’’or “the stress of
migration”…are inaccurate, because these processes
occur on predictable schedules and an individual can
make necessary preparations’. In a broad sense, one
might consider reproduction as a stressor, because
reproducing entails activities and costs that make it
more difficult than not reproducing. Under the
narrower definition given above, reproduction or
migration should not automatically be considered
stressful simply because they are difficult. Difficult
conditions, whether they are physical or
psychological, often do not provoke a physiological
stress response unless they are unpredictable or
uncontrollable. The importance of predictability and
control has been shown very neatly in experiments
with rats Rattus norvegicus9. For example, two rats
can be put in a divided cage with a shock grid on the
floor, so that both are subject to the same series of
electric shocks. If one rat can decrease the rate of
shocks by pressing a lever, the rat without the lever
(without control) typically has a larger GC response,
in spite of facing an identical physical challenge.
Social dominance
and stress hormones
Scott Creel
In most cooperatively breeding birds and mammals, reproductive rates are lower
for social subordinates than for dominants, and it is common for reproduction
in subordinates to be completely suppressed. Early research conducted in
captivity showed that losing fights can increase glucocorticoid (GC) secretion,
a general response to stress. Because GCs can suppress reproduction, it has
been widely argued that chronic stress might underlie reproductive
suppression of social subordinates in cooperative breeders.Contradicting this
hypothesis, recent studies of cooperative breeders in the wild show that
dominant individuals have elevated GCs more often than do subordinates. The
findings that elevated GCs can be a consequence of subordination or a cost of
dominance complicate the conventional view of social stress, with broad
ramifications for the evolution of dominance and reproductive suppression.
Scott Creel
Dept of Ecology, Montana
State University,
Bozeman, M 59717, USA.
e-mail:
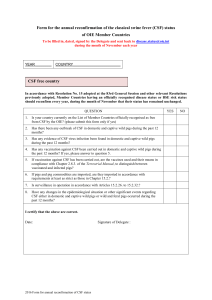
Fig. 1. Examples of agnostic interaction. Agonistic and aggressive
behaviors are similar in most social carnivores, even those that are
distantly related. A winner and loser can be assigned, and used to
estimate social status. (a) African wild dog
Lycaon pictus
greeting by
two subordinates as a dominant individual approaches. The dog in the
foreground is sending mixed signals of dominance (tail up and fanned)
and subordination (play bow). (b) A dominant dwarf mongoose
Helogale parvula
pins a subordinate to the ground as it rolls onto its
back in submission.

Similarly, two rats can be subjected to an identical
series of shocks, but one is given a warning sound
before the shocks so that they are predictable, which
significantly reduces its GC response9.
Because of the impact of predictability and control
on GC responses, behavioral interactions can be
potent stressors (Fig. 1). For over 30 years, it has been
known that aggressive or agonistic interactions can
provoke large and persistent increases in GC
secretion. Influential early work on this issue was
conducted with captive rodents and primates, often
by grouping unfamiliar individuals, observing the
fights that ensued and comparing the GC levels of
winners and losers10–12. In this situation, both
winners and losers showed a strong stress response,
but the response was larger among losers. In these
early studies, losers were generally called
‘subordinates’, and winners were called ‘dominants’.
These studies are the origin of the conventional view
that social subordination is stressful.
Winner–loser contests and naturally formed
hierarchies
These were ground-breaking studies that revealed
much about the associations between aggressive
behavior and stress responses. However, such studies
do not necessarily reveal the consequences of living as
a subordinate in a social group that has a settled
dominance hierarchy. The rate and severity of
fighting are high immediately after strangers are
grouped, particularly among males, which were the
focus of most of the winner–loser studies.
TRENDS in Ecology & Evolution
Vol.16 No.9 September 2001
http://tree.trends.com
492 Opinion
Table 1. Relationships between basal glucocorticoid levels and social status within cooperatively breeding groupsa,b
Species Sex Basal GC Social Reproductive Captive Method of Notes Refs
pattern system suppression or wild sampling GCs
of subordinatescstudy
Rodents
Naked mole-rat F and M Sub > Dom Social and High skew Captive Urine Long-term studies 30
Heterocephalus glabe
cooperative
Alpine marmot M Dom > 3 types, Social and Low skew Wild Blood: long and Categorized subs by 24
Marmota marmota
of sub cooperative variable lag yearling–adult and
Dom < 1 type from trapping son–nonson
of sub to sample
Primates
Common marmoset F Dom > Sub Social and High skew Captive Blood 28
Callithrix jacchus
cooperative
Black tufted-ear F Dom = Sub Social and High skew Captive Urine 33
marmoset cooperative
Callithrix kuhli
M Dom > Sub Social and High skew Captive Urine Additional data from
cooperative J. French
Ring-tailed lemur F Dom > Sub Social and Low skew Wild Feces 25
Lemur catta
cooperative
Cotton-top tamarin F Paired > Sub Social and High skew Captive Urine 34
Saguinus oedipus
with M cooperative
Birds
White-browed F and M Dom = Sub Social and Low skew Wild Blood Very low GC levels for 21
sparrow weaver cooperative all ranks
Plocepasser mahali
Florida scrub jay F Dom > Sub Social and Low skew Wild Blood During all nest stages 22
Aphelocoma
cooperative
coerulescens
M Dom = Sub Social and Low skew Wild Blood Dom > Sub at mating 35
cooperative stage
Harris' hawk F and M Dom = Sub Social and High skew Wild Blood NS ANOVA for GC in 35
Parabuteo unicinctus
(see notes) cooperative breeders, auxiliaries
and juveniles
Carnivores
Wolf F and M Dom = Sub Social and High skew Captive Blood In wild packs, 36
Canis lupus
cooperative Dom > Sub
Dwarf mongoose F Dom > Sub Social and High skew Wild Urine Acute GC: Dom > Sub 14,26
Helogale parvula
cooperative
M Dom = Sub Social and High skew Wild Urine Acute GC: Dom < Sub
cooperative
African wild dog F and M Dom > Sub Social and High skew Wild Feces 15,26
Lycaon pictus
cooperative
aThis excludes studies in which individuals in different groups or on different territories are compared, for example comparisons of unmated, monogamously mated and
polygynously mated male birds on neighboring breeding territories.
bAbbreviations GC, glucocorticoid; NS, not significant at
P
= 0.05.
cHigh skew refers to species in which reproductive success is highly skewed within groups (subordinates rarely breed); Low skew, species in which subordinates often breed.

For example, Blanchard et al.13 put 100 male rats in
a system of clear tunnels and recorded their behavior,
along with changes in body mass and plasma GC levels.
Initially, rates of fighting exceeded 40 fights per hour,
for both dominants and subordinates. After 13 days,
subordinates had an average of 17 body wounds, and
had lost more than 20% of their initial body mass, in
spite of being removed from the colony on four days to
feed for eight hours. Aggression this severe would be
unusual for a social species in the wild, where agonistic
encounters rarely escalate to the point of wounding
and rates of aggression are lower14,15. Indeed, it is
generally thought that social dominance evolves to
avoid the costs and risks of escalated fights, when the
outcome can be reliably predicted16. Perhaps work on
GC responses of winners and losers could be related to
the social events that follow immigration or group
formation in the wild, when social relationships are
often established through fighting17–19.
Captive winner–loser studies do not necessarily
predict relationships between social status and basal
stress hormone levels for stable social groups in the wild,
because of substantial differences in social context. A set
of recently paired males in a small enclosure differs in
many ways from a wild group (often formed of relatives)
that has formed by recruitment of offspring over many
years, with individuals free to disperse if they choose.
Captive subordinates cannot avoid dominant
individuals as effectively as they would be able to in the
wild. In the wild, moving away is a common means of
terminating an attack if behavioral appeasement does
not work. Of course, the ultimate form of moving away
from dominant individuals is dispersal, an option not
open to captive animals. In spite of these complexities,
captive winner–loser studies are the original basis of
the common argument that the ‘stress of
subordination’or ‘psychological castration’might
underlie reproductive suppression among
subordinates in cooperatively breeding species20–22.
Given this, it is interesting to ask what recent studies
reveal about the endocrine consequences of rank in
social species, particularly in cooperative breeders.
TRENDS in Ecology & Evolution
Vol.16 No.9 September 2001
http://tree.trends.com
493
Opinion
TRENDS in Ecology & Evolution
9
5
8
7
6
4
3
1
2
0D > S D > SS > D S > DD = S D = S
Relationship between rank and GCs
Number of cases
Fig. 2. The relationship between basal glucocorticoid (GC) levels and social
status in species with cooperative breeding (purple bars) and in other
social systems (yellow bars). D and S represent dominant and subordinate:
D>S means that GC levels were significanty higher in dominant
individuals, D =S means that GC levels are not detectably affected by rank,
and S >D means that GC levels were significantly higher in subordinates.
Table 2. Relationships between basal glucocorticoid levels and social status within groups that do not breed cooperatively, and in
winner–loser studiesa,b
Species Sex Basal GC Social Reproductive Captive Method of Notes Refs
pattern system suppression or wild sampling GCs
of subordinatescstudy
Rodents
Mouse
Mus musculus
M Sub > Dom Solitary No Captive Blood Winner–loser studies 10,11
ancestral
Rat
Rattus norvegicus
M Sub > Dom Solitary No Captive Blood Short- and long-term 37
ancestral studies
Primates
Squirrel monkey M Sub > Dom Social No Captive Blood See next study 12,38
Saimiri sciureus
M Dom > Sub Social No Captive Blood See previous study
Rhesus macaque M Dom = Sub Social No or low skew Captive Blood 0.3 Ha enclosure 39
Macaca mulatta
(enclosure)
Lesser mouse lemur M Dom = Sub Solitary Low skew Captive Blood 40
Microcebus murinus
Olive baboon M Sub > Dom Social Low skew Wild Blood Only with stable 2,32
Papio anubis
hierarchy
Birds
Harris’ sparrow F and M Sub > Dom Summer pairs, No Wild Blood 41
Zonotrichia querula
winter flocks
White-throated sparrow F and M Sub > Dom Summer pairs, No Wild Blood 42
Zonotrichia albicollis
winter flocks
Suids
Domestic pig F Sub > Dom Solitary, No or low skew Captive Saliva Dom/Sub = ‘high–low 43
Sus scrofa
ancestral success’
aThis excludes studies in which individuals in different groups or on different territories are compared, for example comparisons of unmated, monogamously mated and
polygynously mated male birds on neighboring breeding territories.
bAbbreviations GC, glucocorticoid; NS, not significant at
P
= 0.05.
cHigh skew refers to species in which reproductive success is highly skewed within groups (subordinates rarely breed); Low skew, species in which subordinates often breed.

Endocrine correlates of rank in social species
Tables 1 and 2 summarize 25 studies that relate basal
GC concentrations to rank. These studies used
variable methods, which probably affected their
results to some (unknown) degree. For cooperative
breeders, the list is complete. Of the 25 studies, eight
found that basal GC levels were significantly higher
among subordinates, eight found that GC levels were
significantly higher among dominants, and eight
failed to detect an effect of social status. Based simply
on the number of studies that have found each broad
pattern, there is often an association between basal
GCs and rank (64% of the studies), but it was equally
common for dominants and subordinates to be the
class in which basal GC levels are elevated.
The eight cases in which dominants had the higher
basal GCs share one obvious similarity: all were
cooperative breeders (Fig. 2). Five of these studies
were conducted with naturally assembled groups in
the wild. By contrast, only one of the eight cases in
which subordinates had higher GC levels came from a
cooperative breeder, and five of these studies were
conducted in captivity. Considering cooperative
breeders only, GCs were higher in dominants in eight
cases (nine if the cotton-top tamarin Saguinus
oedipusis included, where subordinates were
compared to monogamously mated females), equal in
six cases, and higher in subordinates in two cases.
(This includes olive baboons Papio anubis; if one
classifies baboons as social but not cooperative,
then GCs were higher insubordinates only for
captive naked mole rats Heterocephalus glaber
among the cooperative breeders.) Narrowing
further still to studies of cooperative breeders in
the wild, five cases support the hypothesis that
it is stressful to be dominant, whereas one
supports the hypothesis that it is stressful to be
subordinate.
TRENDS in Ecology & Evolution
Vol.16 No.9 September 2001
http://tree.trends.com
494 Opinion
When a difference exists between dominants and subordinates in
circulating glucocorticoid (GC) concentrations, it is possible that
the difference is not a result of social status itself, but of some
other trait that varies among individuals of different ranks. Such
traits might also vary among individuals of similar rank, and
several studies have noted that dominants or subordinates of
different behavioral types differ in their basal GC levelsa–c. For
example, Blanchard
etal
.dfound that, in comparison to dominant
rats, subordinates had higher basal GC concentrations and lower
acute GC levels in response to the stress of restraint. They then
ranked subordinates by the strength of their acute GC response,
classifying subordinates as ‘responders’ or ‘nonresponders’, and
showed that there was a pronounced difference between the two
groups in acute GC response, as shown for simulated data in
Fig. Ia. They then demonstrated that responders and
nonresponders differed significantly in some aspects of behavior.
From Fig. Ia, it appears obvious that there are fundamental
differences between nonresponders and responders: they are
two distinct types, and the underlying frequency distribution of
acute GC concentrations is bimodal. This is not a safe conclusion.
For these data, the apparent existence of two types of
subordinates is entirely a result of the process of
post-hoc
classification. The underlying frequency distribution is a
Gaussian distribution with
µ
=4 and
σ
=2, from which 100 random
values were drawn (Fig. Ib) to create the data for Fig. Ia. Through
post hoc
separation of the left and right halves of the distribution,
it appears that two classes of subordinates exist (Fig. Ia), when
the distribution of GC responses is actually unimodal.
This example shows that the conclusion that several types or
‘styles’ of dominant or subordinate exist is sometimes not well
supported by data. Of course, this does not mean that multiple
types do
not
exist. For example, Virgin and Sapolskybclassified
subordinate baboons on the basis of behavioral traits, rather than
making a
post hoc
classification on the basis of GC levels, and
found that some traits were good predictors of acute GC
responses (but were not good predictors of basal GC levels).
References
a Kotrschal, K. et al. (1998) The relationship between social stress and
dominance is seasonal in greylag geese. Anim. Behav. 55, 171–176
b Virgin, C.E. and Sapolsky, R.M. (1997) Styles of male social behavior and
their endocrine correlates among low-ranking baboons. Am. J. Primatol. 42,
25–39
c Mendl, M. et al. (1992) Phsyiological and reproductive correlates of
behavioural strategies in female domestic pigs. Anim. Behav. 44, 1107–1121
d Blanchard, D.C. et al. (1995) Visible burrow system as a model of chronic
social stress: behavioral and neuroendocrine correlates.
Psychoneuroendocrinology20, 117–134
Box 1.Types of dominants and subordinates
6.5
Glucocorticoid concentration
(ng g–1 dry feces)
5.5
4.5
3.5
2.5
1.5 Nonresponder Responder
Post-hoc
classification
24
I (a)
(b)
Number of observations
20
12
16
8
4
0–10123456789
Glucocorticoid concentration (ng g–1 dry feces)
TRENDS in Ecology & Evolution

From Tables 1 and 2, (two) general conclusions can
be drawn. First, the relationship between basal GCs
and rank is highly variable. Variables that might
affect the strength and direction of this relationship
include the social system, domestication, phylogeny,
the behavioral traits that are associated with high and
low rank, and whether the study was conducted in the
wild or in captivity (Box 1). Second, although the data
are still sparse for the purposes of comparative
analysis, it is uncommon for subordinates of species
that live in permanent groups to experience
chronically elevated GCs (Fig. 1). Only one study of a
social species in the wild has found elevated basal GCs
in subordinates23, whereas five field studies have
found higher levels in dominants. These five cases are
phylogenetically well distributed, including a bird
(Florida scrub jay Aphelocoma coerulescens)22, a
rodent (alpine marmot Marmota marmota)24, a
primate (ring tailed lemur Lemur catta)25 and two
carnivores (dwarf mongoose Helogale parvula and
African wild dog Lycaon pictus)15,26.
The interpretation of differences among ranks in
GC levels is not entirely clear-cut. Apriori, the class of
individuals with higher basal GCs would generally be
considered more socially stressed. However, data from
captive common marmosets27,28 show that GC levels
do not increase upon attaining dominance: rather, GC
levels decrease among subordinates, in parallel with
estrogen levels. Field studies have not generally tested
whether the differences between ranks arise through
increased GC secretion in one class or reduced secretion
in the other class, although limited data from African
wild dogs and dwarf mongooses show that GC levels
increase upon attaining dominance. Considering the
impact of GCs on fitness, the difference in mechanisms
might be moot, if the social class with higher basal GC
levels is exposed to more of their harmful effects.
Implications and future research
If dominant animals do generally have elevated GC
levels among cooperative breeders, this would
represent a major shift in our view of social stress.
We must consider the stress of domination, as well as
the stress of subordination, and how these stressors
might affect social evolution. There are several
interesting questions to address. First, what
behavioral differences among species (Box 2)
predict whether stress will fall more heavily on
dominants or on subordinates (or perhaps on mid-
ranking animals, in some cases)? This is already an
active field of study25,29, and two possibilities are that
dominants have elevated GCs when a hierarchy is
unstable2 or in species in which dominants fight more
often than do subordinates26.
Second, what are the nonGC-mediated
mechanisms by which rank affects sexual behavior
and sex steroid levels? It is already clear that
reproductive suppression is not mediated by
chronic GC elevation for most of the cooperative
breeders that have been studied (Table 1, species
listed as ‘high skew’). However, many aspects of
reproductive behavior and sex-steroid secretion
are depressed in subordinates of these
species14,15,22,30. Considering the broad range of
pathologies that chronic GC elevations can cause2,4,
it is perhaps not surprising that reproductive
suppression is rarely GC mediated in species for
which reproductive suppression is a normal
feature of social organization. With such a social
system, stress-mediated mechanisms of
suppression would be evolutionarily invasible by a
direct mechanism of suppression that did not
involve GC elevation, and thus avoided the costs of
immune suppression and other pathologies. If this
line of reasoning is correct, stress-mediated
TRENDS in Ecology & Evolution
Vol.16 No.9 September 2001
http://tree.trends.com
495
Opinion
Although the terminology is cumbersome, there is an important
distinction between short-term (‘acute’) and long-term (‘chronic’)
stress responses. When exposed to a stressor, a typical individual
will mount a pronounced glucocorticoid (GC) response within
two to ten minutes. During this acute response, circulating GC
levels typically rise several fold, but drop back to basal levels
minutes (or perhaps a few hours) after the stressor is removed.
This short-term response is generally considered adaptive,
because it shifts energy and resources away from physiological
processes that can be curtailed briefly without harm, putting
these resources instead toward resolving the stressful condition.
By contrast, if an elevation of GCs persists for days or weeks,
then many of the short-term benefits become long-term pathologies,
including immune suppression, reproductive suppression, gastric
ulcers and muscle wasting. Sapolskyaprovides an excellent review
of the short-term benefits and long-term harms that elevations of
GCs usually provoke, and their mechanisms. Because of the
dichotomy in the effects of acute and chronic GC responses, studies
of social stress generally focus on chronic elevations of GCs
(elevated basal values). Some studies go further by also testing
for effects of social status on acute GC responsesb–d. When basal
GC concentrations are elevated, a common consequence is a
weakening of the acute GC responsed, probably through changes in
negative feedback mechanisms in the hypothalamic–adrenal–
pituitary axis. This is an endocrine vicious circle: a chronically
high baseline can provoke pathologies, and a weak acute
response is ineffective at handling short-term stressorse.
References
a Sapolsky, R.M. (1992) Neuroendocrinology of the stress response. In
Behavioral Endocrinology (Becker, J.B. et al., eds), pp. 287–324,
Massachusetts Institute of Technology Press
b Schoech, S. et al. (1991) Reproductive endocrinology and mechanisms of
breeding inhibition in cooperatively breeding Florida scrub jays
(Aphelocoma c. coerulescens). Condor93, 354–364
c Creel, S. et al. (1996) Social stress and dominance. Nature379, 212
d McEwen, B.S. and Schmenk, H.M. (1994) The Hostage Brain, Rockefeller
University Press
e Virgin, C.E. and Sapolsky, R.M. (1997) Styles of male social behavior and their
endocrine correlates among low-ranking baboons. Am. J. Primatol. 42, 25–39
Box 2.Chronic and acute stress
 6
6
 7
7
1
/
7
100%