Surg Radiol Anat (2010) 32:409–414
DOI 10.1007/s00276-009-0546-3
123
ANATOMIC VARIATIONS
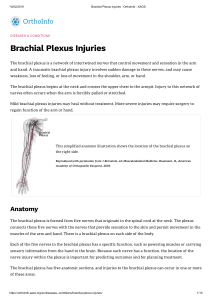
Anatomical variations of lumbosacral plexus
Viktor Matejbík
Received: 14 April 2009 / Accepted: 6 August 2009 / Published online: 21 August 2009
© Springer-Verlag 2009
Abstract
Basis Deviations detected during spinal operations have
motivated us to start research related to variations of lum-
bosacral plexus formation. Aim of this work was to Wnd out
deviations of its formation from ascension of particular
roots from foramen invertebrale and foramina sacralia up to
formation of terminal branches.
Set One hundred lumbosacral plexi have been examined
in 50 adult cadavers for a purpose to Wnd out an incidence
of neural variations. We have observed participation of
Th12 root, L4 and L5 roots in its formation, as well as vari-
ous deviations from ascension of particular plexiform roots
up to their ending branches. For lumbal plexus, we have
observed four nerve roots and six lumbal nerves; for sacral
one, three sacral roots with a share of S4 and lumbosacral
trunk formed of L4 and L5 roots and four sacral nerves. We
have considered also their course, anastomoses and thick-
ness. We highlight motoric innervation particularities in
relation to diagnostics besides anatomical complexity and
variability.
Results Variations on the level of neural roots were com-
mon, nerve formations were uncommon. Thickness of neu-
ral roots, formation or their absence were dependent on the
type of plexus, particularly in sacral area. For lumbal
plexus, L1 root was the thinnest and L4 root was the thick-
est. L3 root was the thickest in six cases. Fifth lumbal root
usually completely Wlled foramen invertebrale. We have
observed double ascension of L4 root from foramen inver-
tebrale in 25 cases and plexiform in Wve cases. In other
cases it was branched in various distance following ascen-
sion from foramen invertebrale. Plexiform ascension of L3
root along with L4 root was present in two cases. Double
ascension of L3 root was present in four cases. L2, L3 along
with L5 roots were doubled in two cases. Iliohypogastric
nerve was the longest, ilioinguinal was the thinnest and ner-
vus femoralis was the thickest. Changes in sacral plexus on
the level of neural roots have been observed 41-times. Dou-
ble ascension of L5 root was present in eight cases and plex-
iform in four cases. Double S1 root at ascension from
foramina sacralia was present 16 times, S2 8 times, S3 once
and S1 along with S2 4 times. S1, S2 and S3 roots were
branched in various distance following ascension from
foramina sacralia in 15 cases. Truncus lumbosacralis was
thickened in 19 cases, a share from L4 root was thicker as
L5 root in 11 cases. Low level of connection between trun-
cus lumbosacralis and S1 root was observed in 10 cases.
Nervus ischiadicus has branched into tibial and peroneal
portions already in minor pelvis in two cases. The level of
distance of n. (nervus) gluteus superior, n. gluteus inferior,
n. cutaneus femoris posterior and n. pudendus was depen-
dent on the plexus type.
Conclusion This study enabled us to Wnd out and to
describe extraordinary anatomical deviations in formation
of neural roots and nerves of lumbal and sacral plexus,
undescribed yet.
Keywords Lumbosacral plexus · Sacral plexus · Lumbal
plexus · Anatomical variations
Introduction
Lumbal plexus is formed by ventral branches of L1–L4 in
musculus psoas major with a share of Th12 or without it. It
V. Matejbík (&)
Department of Neurosurgery, Derer’s Faculty Hospital,
Comenius University, Limbova 5, 833 05 Bratislava,
Slovak Republic
e-mail: matejcik@pobox.sk

410 Surg Radiol Anat (2010) 32:409–414
123
is located on internal surface of dorsal abdominal wall, ven-
trally from processus transversus of coxal vertebrae [1, 2].
It is protected with layers of the muscle and bone struc-
tures. As well-protected and safely located structure, it pro-
vides better safety to the plexus. Lumbosacral plexopathies
are therefore less frequent peripheral lesion aVecting lower
extremities.
Sacral plexus is formed by ventral branches of S1–S3
with a share of S4 and very important lumbosacral trunk
formed of L4 and L5 roots. It extends above sacroiliac junc-
tion and consequently connects with sacral nerves forming
n. ischiadicus.
Depending of Th12 root presence, we say on high cranial
preWxed plexus or low caudal one. Boundary root partici-
pating in the plexi formation is L4 root, which in some
cases is largerly involved in lumbal plexus formation and
sacral in others. Its proportion in that or the other plexus
helps to characterize the whole plexus—as cranial preWxed
with a share or caudal postWxed without a share of Th12.
It is necessary to realize that L4 root does not play,
always, the role of boundary root; sometimes it is L5 root
[3].
As for literature available for us, we have met only with
orphan works devoted to these problems [1–8].
Materials and methods
The study is related to 50 fresh cadavers without innate or
detected abnormalities, tumour diseases, orthopaedic defor-
mities and spinal operations. Ethics committee approval
was obtained prior to the study. However, to the best of our
literature search, we could not come across any study per-
formed by the use of those reference structures or points.
Therefore, we were not able to discuss our Wndings on the
topography lumbosacral plexus and roots with the results of
others. Lumbosacral plexi were clariWed in all of them,
bilaterally from anterior access following evisceration.
Lumbal plexus was clariWed following thorough dissection
of m. psoas major.
We have observed extraforaminal deviations of particu-
lar root formation up to their ending branches, and partici-
pation of Th12 and L4 roots, L5, respectively, in its
formation.
Main lumbal neural roots were localized along with the
surface of the most dorsal part of vertebral bodies and inter-
vertebral discs.
Lumbal plexus is formed laterally from intervertebral
foraminas and passes through m. psoas major. Each lumbal
root ascends through superior part of corresponding inter-
vertebral foramina. Ascending from foramina, it extends
downward along with lateral surface of caudal pedicle.
Here it is crossed by intertransversal ligament with an aim
to maintain ventrolateral routing. Most of neural sleeves are
anchored on cranial pedicle and only small space between
lower bound of pedicle and neural roots is present.
Minor motoric branches are depleted directly in m. psoas
major, while longer ones leave the muscle on various sites
and extend aslope downward into pelvic area and leave it
under ligamentum inguinale. N. obturatorius leaves the pel-
vis through foramen obturatorius.
Lumbal nerves—n. iliohypogastric, n. ilioinguinal, n.
genitofemoral, n. cutaneus femoris lateralis, n. obturatorius,
n. femoralis and sacral nerves—n. ischiadicus, n. gluteus
superior, n. gluteus inferior, n. cutaneus femoris posterior
and n. pudendus, were identiWed bilaterally.
Nerves of sacral plexus are located on anterior area of
m. piriformis. Branches of iliac vessels are in the vicinity of
S1, S2 and S3 roots.
Results
As with upper extremity, neural trunks of lower extremity
in high cranial form also receive more fascicles from above
located nerves in comparison with low caudal form.
Th12 and L1 roots are thicker in preWxed type. Then L4
root contributes more signiWcantly to sacral plexus and S3
and S4 roots are absent. If L4 root does not contribute to
sacral plexus or contributes only minimally, L5 root is
thiner, S1–S3 roots are thicker and S4 root is present too. L5
Table 1 Variations of lumbal plexus roots
Number of L
plexus root
Double root
ascension
Plexiform
root ascension
L2 2 –
L3 4 –
L4 25 5
L3 + L4 – 2
L2 + L3 + L5 2 –
Total 33 7
Table 2 Variations of sacral plexus roots
Number of S
plexus root
Double
root ascension
Plexiform
root ascension
L5 8 4
S1 16 –
S2 8 –
S3 1 –
S4 – –
S1 + S2 4 –
Total 37 4

Surg Radiol Anat (2010) 32:409–414 411
123
root is the largest nerve contributing to sacral plexus in
postWxed type. When determining the type of plexus, the
most appropriate was L4 root, eventually also L5 root.
PreWxed type has been occured in 19 cases, postWxed in
Wve cases. Variations in the area of plexi were most fre-
quently on the level of neural root formation (Tables 1, 2).
Foramen intervertebrale is the narrowest on the level of
L5/S1. It was usually completely Wlled with Wfth lumbal
root. L4 root participated in truncus lumbosacralis forma-
tion in every case. L5 root did not contribute to lumbal
plexus in no case. The branch from L4 root to L5 root was
thicker as L5 root itself in 11 cases (Fig. 1). Thin connec-
tion of L4 root with L5 root was present in majority of
cases. Thick truncus lumbosacralis was present in 19
cases (Figs. 2, 3). We have observed junctions of L3 root
to L4–L5–S1 roots in 12 cases.
Double ascension of roots in lumbal plexus was
observed in 33 cases and plexiform in seven cases.
Among them, three roots of L2, L3, L5 simultaneously in
two cases (Fig. 4). Double ascension of L2 root was
observed in two cases, L3 in four cases, L4 in 25 cases. For
other cases, L4 root was branched in various distance fol-
lowing ascension from foramen intervertebrale. Plexiform
ascension of L4 root was observed in Wve cases, and L3
along with L4 roots in two cases. L1 root was the thinnest
and L4 was the thickest root of lumbal plexus. Iliohypogas-
tric nerve was the longest, ilioinguinal nerve was the thin-
nest and n. femoralis was the thickest. We have observed
most deviations of its formation in its case. Shares of L2, L3
roots were dominant in 40 cases and L4 share was minor,
also from L1 root in 1 case. Common trunk formation of n.
femoralis may be more distal, must not be formed respec-
tively, six cases. L3 root was the thickest in six cases.
Participation of L4 root in sacral plexus formation was
observed in every case. Absence of junction between the
plexi was not observed in any case. In cases of preWxed
plexus—thick truncus lumbosacralis in sacral plexus for-
mation, only S1 and S2 roots were present and nerves of
sacral plexus ascended from L5, S1 and S2 roots.
For postWxed type, truncus lumbosacralis was thin, S1,
S2 roots were thicker (Fig. 5). S3 and S4 roots were present
too. For other cases, truncus lumbosacralis and S1, S2, S3
Fig. 1 PreWxed type of
lumbosacral plexus, right side,
front view, in craniocaudal
direction, surrendered branches
of the spine from left to right:
a branch from L4 root (1) is
thicker than L5 root (2), thick
truncus lumbosacralis (3),
S1 root (4), S2 root (5)
Fig. 2 Thick truncus lumbosac-
ralis, left side, front view, in cra-
niocaudal direction, from right
to left: thin S1 and S2 roots, a
branch from L4 root (1), L5 root
(2), truncus lumbosacralis (3),
S1 root (4), S2 root (5),
S3 root (6)

412 Surg Radiol Anat (2010) 32:409–414
123
roots were present. Changes in sacral plexus on the level of
neural roots were observed 41 times. Double ascension of
L5 root (Fig. 6) was present in eight cases. Plexiform ascen-
sion of L5 root in four cases (Fig. 7). Double ascension of
S1 root following ascension from foramina sacralia
16 times, S2 8 times and S3 once. S1+S
2 roots simulaten-
ously 4 times (Fig. 8). S1, S2 and S3 roots were branched in
various distance following ascension from foramina sacra-
lia in 15 cases. N. ischiadicus was branched into tibial and
peroneal portion already in minor pelvis in two cases.
N. cutaneus femoris posterior leaves S1 root and takes
shares of S2 and S3 roots.
Fig. 3 Thick truncus
lumbosacralis (1), left side, front
view, in craniocaudal direction,
from right to left: thin S1 (2) and
S2 (3) roots branching following
ascension from foramina
sacralia
Fi
g.
4
Dou
bl
e ascens
i
on o
f
L2
and L3 roots, right side, front
view, in craniocaudal direction,
from left to right: L5 root (1), L4
root (2), L3 root (3), L2 root (4)
Fig. 5 PostWxed type of lumbo-
sacral plexus, right side, front
view, in craniocaudal direction,
from left to right: thin branch
from L4 root (1) to L5 root (2),
truncus lumbosacralis (3), S1
root (4), S2 root (5), S3 root (6),
S4 root (7), n. obturatorius (8),
n. femoralis (9)

Surg Radiol Anat (2010) 32:409–414 413
123
N. pudendus was formed depending on the plexus type
from S2 and S3 roots. If main trunk of n. pudendus leaves S3
root, it takes a share of S2 root.
Discussion
Lumbosacral plexus is an analogue of brachial one. DiVer-
ences in lumbosacral plexus formation are demonstrated in
that manner that segmental participation of certain nerves,
as well as their participation in tissue innervation may be
changed, as in brachial one on the periphery.
Nevertheless, interest in details of both plexi formation
is not the same. It is because injuries as well as variations of
lumbosacral plexus formation are less frequent in compari-
son with brachial one. Indications for reconstructive opera-
tions of lumbosacral plexus are exceedingly rare.
There are a lot of variations of standard pattern related to
innervation from lumbosacral plexus, but innervation from
the Wrst two sacral nerves is quite constant. S2 root also
innervates glutei and musculus biceps femoris. N. gluteus
superior innervates musculus gluteus medius—so-called
“deltoid” of coxal joint, musculus gluteus minimus and
extends forward, innervating m. tensor fascia lata, while S3
root usually innervates Xexors of big toe and only rarely
other muscles on the extremity. S4 root innervates no mus-
cles on the extremity.
L4 root contributes to lumbal and sacral plexi and there-
fore it is called branched or furcate “furcal nerve”. With the
exception of L4 root, in the literature we did not Wnd any
cases of root duplication or root fragmentation of lumbosa-
cral plexus that could constitute real operational pitfalls for
neurosurgeon and anaesthetist during regional anaesthesia.
Weber [8] pointed out to some variations of lumbal
plexus. Following Bergman et al. [9], both plexi—lumbal
and sacral—are connected with furcal nerve in 91.8%.
The connection was not found in 7.4%. Iczi et al. [6] has
seen only one case of furcal nerve and had not observed
any variation of the connection between lumbal and sacral
plexus.
Fig. 6 Double ascension of L5
root, left side, front view, in cra-
niocaudal direction, from right
to left: S1 root (1), L5 root (2, 3),
L4 root (4), a branch from L4
root to L5 root (5),
n. obturatorius (6),
n. femoralis (7)
Fi
g.
7
P
l
ex
if
orm ascens
i
on o
f
L5 root, left side, front view, in
craniocaudal direction, from
right to left
Fi
g.
8
Dou
bl
e
S
1 root, rig
h
t
side, front view, in craniocaudal
direction, from left to right:
a branch from L4 root (1), L5
root (2), truncus lumbosacralis
(3), S1 root (4), S2 root (5),
S3 root (6)
 6
6
1
/
6
100%