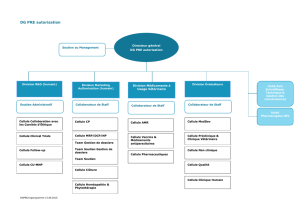
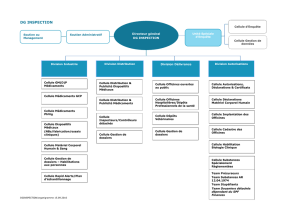
L`équipe intitulée « Individualisation des doses des médicaments

L’équipe intitulée « Individualisation des doses des médicaments anticancéreux » constitue une
nouvelle équipe du Centre de Recherches en Cancérologie de Toulouse (CRCT) INSERM UMR1037,
Université Paul Sabatier, CNRS ERL 5294 localisée à l’Institut Universitaire du Cancer de Toulouse
(IUCT) – Oncopole. Dans sa stratégie de développement pour le prochain quinquennat 2016-2020, le
CRCT souhaite renforcer les activités de recherche de cette équipe en renforçant son personnel de
recherche. Notre équipe souhaite accueillir un(e) chercheur(e)statutaire INSERM ou CNRS ayant une
expérience en analyse de données pharmacologiques (pharmacocinétiques, pharmacocinétiques-
pharmacodynamiques, pharmacogénétiques) selon, en particulier, des approches de population.
Au sein de l’équipe, il sera proposé à ce chercheur de s’impliquer dans la conception, la réalisation et
l’analyse d’essais cliniques de médicaments développés en Onco-Hématologie. Une expérience solide
de l’utilisation de logiciels de modélisation de pharmacocinétique est nécessaire (tels que NONMEM,
Monolix, R…). Le/la candidat(e) sera amené(e) à collaborer sur ces projets avec les différents
membres de notre équipe multidisciplinaire (pharmacologie, pharmacogénétique, biostatistique,
chimie analytique). Il/elle aura accès à toutes les plates-formes et facilités administratives du CRCT
ainsi qu’au recrutement d’étudiants dans le cadre de stages de recherche M2R et doctorants ou post-
doctorants.
Publications de l’équipe illustrant notre domaine de recherche :
- Schmitt A et al: Factors for hematopoietictoxicityof carboplatin: refining the targeting of
carboplatin systemic exposure. J Clin Oncol 2010;28:4568-4574 ;
- White-Koning M et al: Population analysis of erlotinib in adults and children reveals
pharmacokinetic characteristics as the main factor explaining tolerance particularities in
children. Clin Cancer Res 2011;17:4862-4871
- Thomas F et al : Genotyping of a family with a novel deleterious DPYD mutation supports the
pretherapeutic screening of DPD deficiency with dihydrouracil/uracil
ratio.ClinPharmacolTher2015 Aug 11. doi: 10.1002/cpt.210. [Epub ahead of print]
- Chalret du Rieu Q et al: Pharmacokinetic/Pharmacodynamic modeling of abexinostat-
induced thrombocytopenia across different patient populations: application for the
determination of the maximum tolerated doses in both lymphoma and solid tumour
patients. Invest New Drugs. 2014;32:985-94.
- Imbs DC et al: Pharmacokinetics of pazopanib administered in combination with
bevacizumab. Cancer Chemother Pharmacol 2014;73:1189-96.
Contact : Etienne CHATELUT (Responsable de l’équipe) chatelut.etienne@iuct-oncopole.fr

Our team entitled « Dose individualisation in anticancer drugs » has recently become part of the
Cancer Research Center of Toulouse (Centre de Recherche en Cancérologie de Toulouse (CRCT)
INSERM UMR1037, Université Paul Sabatier, CNRS ERL 5294) which is situated in the Institut
Universitaire du Cancer de Toulouse (IUCT) – Oncopole. The CRCT’s global strategy for development
for the next five years (2016-2020) includes the development of the research activities within our
team through the recruitment of additional researchers. We would like to welcome in our team an
INSERM or CNRS researcher with experience in pharmacological data analysis (pharmacokinetics,
pharmacokinetics-pharmacodynamics, pharmacogenetics) in particular using the population PK
approach.
The researcher would be invited to get involved in the design, implementation and analysis of clinical
trials for drugs developed in Oncology and Haematology. A solid experience of PK modelling
software such as NONMEM, Monolix, R...) would be required. The researcher would collaborate on
different projects with the other members of our multidisciplinary team (pharmacology,
pharmacogenetics, biostatistics, analytical chemistry). The researcher will have access to all the
platforms and administrative facilities within the CRCT and the possibility to recruit and supervise
students for masters degree placements, doctoral or post-doctoral positions.
The following publications give an idea of our research area :
- Schmitt A et al: Factors for hematopoietic toxicity of carboplatin: refining the targeting of
carboplatin systemic exposure. J Clin Oncol 2010;28:4568-4574 ;
- White-Koning M et al: Population analysis of erlotinib in adults and children reveals
pharmacokinetic characteristics as the main factor explaining tolerance particularities in
children. Clin Cancer Res 2011;17:4862-4871
- Thomas F et al : Genotyping of a family with a novel deleterious DPYD mutation supports the
pretherapeutic screening of DPD deficiency with dihydrouracil/uracil ratio.
ClinPharmacolTher 2015 Aug 11. doi: 10.1002/cpt.210. [Epub ahead of print]
- Chalret du Rieu Q et al: Pharmacokinetic/Pharmacodynamic modeling of abexinostat-
induced thrombocytopenia across different patient populations: application for the
determination of the maximum tolerated doses in both lymphoma and solid tumour
patients. Invest New Drugs. 2014;32:985-94.
- Imbs DC et al: Pharmacokinetics of pazopanib administered in combination with
bevacizumab. Cancer Chemother Pharmacol 2014;73:1189-96.
Contact : Etienne CHATELUT (Team leader) chatelut.etien[email protected]
1
/
2
100%