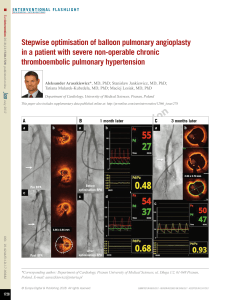
CLI & Popliteal Artery Disease: Endovascular Management
Telechargé par
Rachid Ben Soussia

Critical Limb Ischemia and the Diseased Popliteal
Artery
Mohammad Reza Rajebi, MD,
*
and Constantino Peña, MD
†
The unique anatomical location and particular biomechanical factors affecting the
popliteal artery provide a challenge to determine the proper endovascular, surgical, or
combined intervention for patients with critical limb ischemia who often require prompt
management in the presence of severe lifestyle-limiting symptoms or of the risk of limb
loss or both. Herein, we provide an overview and practical guide for endovascular
management of popliteal artery disease in the setting of critical limb ischemia.
Tech Vasc Interventional Rad 17:170-176 C2014 Elsevier Inc. All rights reserved.
KEYWORDS CLI, popliteal artery disease, peripheral vascular disease, popliteal stents
Introduction
Lower extremity peripheral arterial disease (PAD) is a
common disease with an estimated prevalence of 30% in
patients aged 70 years and older or in those who are 50-69
years of age with a history of cigarette smoking or diabetes.
1
Critical limb ischemia (CLI), defined as rest pain or tissue
loss or both, is the most severe form of lower extremity
PAD. CLI requires immediate medical attention and possi-
ble intervention. The exact contribution of isolated or
concomitant popliteal artery disease in patients with CLI
is unknown, as traditionally superficial femoral artery (SFA)
and popliteal artery diseases are grouped together in the
medical literature. Chronic occlusive arterial disease is the
most common etiology for CLI, while involvement in the
popliteal artery is one of the most challenging vascular beds.
The popliteal artery serves as the single arterial conduit
between the thigh and the calf, presenting a critical inflow
vessel into the calf and the foot. The popliteal artery can have
asignificant variability in diameter from the distal SFA to its
bifurcation into the origin of the anterior tibial artery and the
tibial peroneal trunk. Before any endovascular intervention,
the quality of the popliteal artery proximal and distal to the
lesion must always be considered for possible bypass surgery
in these patients. Additionally, other disease entities such as
popliteal artery aneurysm, entrapment syndrome, cystic
adventitial disease, or trauma can also lead to CLI.
The popliteal artery has traditionally been considered a
“no stent zone”particularly because of the unique bio-
mechanical forces of the area along with the repetitive
motion of the adjacent knee joint. Traditional nitinol stents
had been thought to lack the flexibility, durability, and
conformability necessary. The durability of angioplasty in
this region is extremely variable and likely related to the
morphologic characteristics of the lesions. As studied in
the femoral artery, longer calcified occlusions likely
respond worse to angioplasty than their counterpart does.
The risk of dissection, coupled with the desire not to stent
in this region, has made the popliteal artery an area in
which debulking with atherectomy and the use of remod-
eling balloons (Cryoplasty, AngioSculpt, Chocolate, Vas-
cuTrak, Cutting) have been favored.
Patient Evaluation
The history and physical examination in patients with CLI
is important in determining eventual treatment options.
The quality of the rest pain, relieving and exacerbating
factors, and the other accompanying signs and symptoms
should be questioned. The existing ulcers should be
investigated, and the etiology of ulceration should be
determined. The arterial ulcers usually occur in the toes.
They are painful and appear dry and pale. Given the
multiplicity of medical conditions in these patients, one
should also pay special attention to other diseases that may
confound or overlap with CLI, such as diabetic neuro-
pathy. It is important at the time of angiography to
correlate the patient's symptoms with the existing vascular
disease to determine the revascularization game plan.
170
1089-2516/13/$ - see front matter &2014 Elsevier Inc. All rights reserved.
http://dx.doi.org/10.1053/j.tvir.2014.08.005
*Department of Radiology, Mayo Clinic, Rochester, MN.
†Miami Cardiac and Vascular Institute, Miami, FL.
Address reprint requests to Constantino Peña, MD, Miami Cardiac and
Vascular Institute, 8900 North Kendall Drive, Miami, FL 33176.
E-mail: [email protected]

Endovascular, open surgical, and combined options
should be considered as they apply to the specific patient's
symptoms and angiographic findings. In the setting of
multilevel disease, the treatment of the popliteal artery,
when involved, can be critical.
The involvement of the popliteal artery disease in patients
with CLI has been studied. A landmark study by Graziani et
al evaluated and morphologically categorized diabetic sub-
jects with ischemic foot ulcers. In 417 consecutive patients
who underwent angiography, there were nearly 3000
lesions. More than 50% of the lesions were occlusions, of
which 75% were discovered in the tibial vessels. Of the
patients, 85% had popliteal or femoral-popliteal involve-
ment.
2
A similar study from the United States with a series of
more than 450 patients found most patients with CLI having
occlusions of the popliteal or tibial arteries.
3
Indications for the Procedure
Unlike the patients with claudication, patients with CLI
require an intervention for limb salvage or to decrease the
extent of amputation. Approximately 40% of patients with
CLI who are not able to undergo revascularization will
requireamputationinthenext6months.
4
As previously
mentioned, CLI usually presents a challenge given the
multilevel disease and patient's comorbidities. There are
2 major considerations in revascularization of patients with
CLI. If tissue loss, ulcer, or gangrene is present, the goal of
revascularization is to increase the blood flow to the lower
extremity to provide an acceptable tissue perfusion, which is
required for the healing process. The required blood flow for
tissue repair is more than the flow needed to maintain the
intact healthy tissue of the extremity. However, the long-term
patency is of less importance in this critical setting. In the
presence of rest pain without tissue loss or after the healing of
ulcers, the main purpose of the intervention should be
improving the patient's symptoms, requiring additional
concern about the long-term patency of the diseased vessel.
Involvement of the popliteal artery is important in both
clinical situations. Patency of the popliteal vessel is essential
to provide inflow into the tibial and the pedal vessels.
Procedural Steps
The patient should be positioned supine on the fluoro-
scopy table. Antegrade access in the ipsilateral common
femoral artery is preferred when possible as it provides
better support for the catheters and wires during difficult
interventions, decreases the fluoroscopy time to cross the
diseased aortic bifurcation and tortuous iliac vessels, and
requires shorter catheters and wires. Conversely, antegrade
access is more cumbersome and carries a higher risk in
obese patients. Furthermore, the use of closure devices is
not approved for this access.
With antegrade access, the patient's inguinal region as
well as the ipsilateral abdominal lower quadrant should be
prepared in advance. The access site on the skin is usually
more superior to the inguinal region. If excessive
abdominal fat is present, the hand that holds the access
needle is also responsible for displacing the fat superiorly
with its lateral margin. This helps to create a proper angle
and to avoid unnecessary soft tissue passage. It is impor-
tant to identify the actual arterial access site with fluoro-
scopy. Usually the distal one-third of the femoral head is
an appropriate landmark for the arterial puncture site.
Ultrasonography is a critical component of our practice in
establishing a proper arterial access.
After identifying the proper arterial access site on the
common femoral artery with fluoroscopic and ultrasono-
graphic guidance, a secure arterial access is established.
Initial angiographic evaluation of all of the lesions is
important as discussed previously. This provides the
angiographic information to assess the number, type,
and locations of the obstructing lesions as well as to
recognize the distal and proximal edges, identify the
collaterals, and evaluate the distal runoff along with the
strength and collateralization pattern within the foot.
Revascularization Options
Revascularization of a stenotic or occluded segment within
the popliteal artery follows traditional endovascular princi-
ples. Moreover, intraluminal recanalizations are essential to
avoid subintimal or reentry passages within the middle and
the distal popliteal artery. Subintimal recanalizations within
the popliteal artery increase procedural complications,
particularly at the level of the knee joint as well as near
the bifurcation into the tibial vessels, as they may cause
occlusion of vessel branches or dissections. Additionally, a
subintimal path limits the opportunity to perform atherec-
tomy. However, maintaining an intraluminal access may be
practically impossible in some lesions, especially given the
presence of a hard cap in the proximal end of the lesion.
Retrograde pedal or tibial access may be helpful in these
situations to maintain an intraluminal path. Nevertheless,
when a subintimal recanalization is performed, maintain-
ing a small proximal loop with the hydrophylic guidewire
is crucial to control the length of the subintimal dissection.
Additionally, reentry devices are available to not only
address the technical challenge of reentry but also help
control the location where reentry occurs, allowing the
protection of the middle and the distal popliteal artery if it
is patent. Surgical bypass is generally considered the
treatment of choice for lesions involving the middle and
the distal popliteal artery if the patient is a surgical
candidate and there is sufficient venous conduit.
Traditionally, balloon angioplasty is the primary endo-
vascular treatment for the popliteal artery lesions (Fig. 1).
Similar to the femoral artery, the use of plain old balloon
angioplasty (POBA), particularly for short, noncalcified
lesions, appears to have similar patency, target lesion
revascularization (TLR) rates, and symptom improvement
scores when compared with nitinol stent placement if the
vessel responds well to the initial angioplasty. This was
demonstrated in a study that included 246 patients with
popliteal artery stenosis who were randomized to POBA
CLI and the diseased popliteal artery
171

(127 patients) or nitinol stent placement (119 patients).
The study included 21% patients with CLI and 33%
patients with popliteal occlusions. The average popliteal
lesion measured 42 mm. Highlighting the risk of an
acutely unsuccessful angioplasty requiring stent placement
in the popliteal artery, 25% of patients in the POBA group
crossed over to receive nitinol stent placement because
of dissection or residual stenosis greater than 30%.
Figure 1 A 60-year-old man with a nonhealing ulcer of the left fifth toe (Rutherford category 5). (A) Digital
subtraction angiogram (DSA) demonstrates a long segment occlusion of the popliteal artery and the tibioperoneal
(TP) trunk with reconstitution of the proximal posterior tibial artery (PTA) (arrowhead) and the peroneal artery
(arrow). (B) The lesion was crossed with the aid of a wire and a catheter, and the TP trunk and the PTA were treated
with a 2.5-mm 120-mm balloon catheter (arrow). (C) The popliteal artery was treated with a 4-mm 40-mm
balloon catheter (arrow). (D) Postangioplasty DSA demonstrates resolution of occlusion without flow-limiting
dissection.
Figure 2 A 78-year-old woman with a left first toe ulcer (Rutherford category 5). (A) Angiogram demonstrates a short
segment occlusion of the proximal popliteal artery and stenosis of the TP trunk. (B) The lesions were crossed with the aid of
a wire and a catheter, and a 6-mm 150-mm nitinol stent was deployed in the proximal popliteal artery (arrow). (C) The
distal popliteal artery and the TP trunk were treated with a 3-mm 120-mm balloon catheter (arrow). (D) Postprocedural
angiogram demonstrates resolution of occlusion and stenosis without flow-limiting dissection. TP, tibioperoneal.
M.R. Rajebi and C. Peña
172

Interestingly, based on the actual treatment received,
patients who underwent POBA had similar results when
compared with those who underwent provisional nitinol
stenting (failing POBA first) as well as those who under-
went primary nitinol stenting.
5
In case of unsatisfactory
response to angioplasty, heavily calcified severe lesion,
or a flow-limiting dissection, stent implantation can be
used for restoration of proper blood flow, especially in
the supragenicular segment of the popliteal artery
(Fig. 2).
6
The opportunity to perform a safer and more effective
angioplasty has led to the creation of specialty angioplasty
balloons that aim at reducing dissections and improving
luminal diameter by scoring the plaque or controlled dilata-
tion of the vessel. These include the Chocolate balloon (Cordis
[J&J], NJ), AngioSculpt balloon (AngioScore, Inc, CA)
(Fig. 3), and VascuTrak balloon (CR Bard, AZ) (Fig. 4)among
others. However, there are no randomized trials comparing
the true value of these balloons in the popliteal artery.
Debulking of the popliteal artery lesions with the use of
atherectomy has been used successfully as a method to
improve the vessel lumen and flow. In a study that compared
atherectomy with balloon angioplasty, investigators concluded
that both techniques offer similar midterm patency, limb
salvage, and freedom from intervention despite a discrete
pattern of complications, with dissection being more common
with angioplasty vs a higher rate of thromboembolic events
with atherectomy.
7
Additionally, atherectomy may be an
option to adapt the vessel wall so that it can better respond
to angioplasty (Fig. 5).
As drug-eluting technology becomes more popular, the
use of drug-eluting balloons may help increase the patency
of popliteal artery angioplasty, whereas drug-eluting stents
may increase the patency of those lesions requiring
Figure 3 An 84-year-old woman with rest pain involving the right lower extremity (Rutherford category 4). (A) Angiogram
demonstrates a short segment occlusion of the supragenicular popliteal. (B) The lesion was crossed with the aid of a wire
and catheter and treated with a 4-mm AngioSculpt balloon (arrow). (C) Postangioplasty angiogram shows a spiral
dissection involving the treated lesion (arrow). (D) A 6-mm 4-cm drug-eluting stent was deployed in this region.
Figure 4 A 70-year-old man with a necrotic right third toe (Rutherford category 6). (A) Angiogram demonstrates severe
stenosis of the proximal popliteal artery and multifocal stenotic lesions in the distal popliteal artery. (B) The lesions were
crossedwiththeaidofawireandcatheteranda6-mm8-mm drug-eluting stent was deployed in the proximal
popliteal artery (arrow). (C) The distal popliteal artery was treated with a 4-mm 40-mm VascuTrak balloon (arrow).
(D) Postprocedural angiogram demonstrates significant improvement of the lesions without flow-limiting dissection.
CLI and the diseased popliteal artery
173

stenting by reducing the risk of restenosis (Fig. 6). Studies
focusing on these trends as well as the actual effect of drug-
eluting technology on patients with CLI and limb salvage
rates will be helpful. The effect of drug-eluting balloons
after atherectomy to improve patency, particularly in
calcified vessels, has also been studied
8
; however, data
on the popliteal artery are limited.
Self-expanding covered stent technology (VIABAHN,
WL GORE, AZ) has been studied extensively for long
femoral occlusive lesions as well as for the treatment of
popliteal artery aneurysms. The covered stents appear to
have responded well to the constant biomechanical motion
of the popliteal artery. The covered stents with heparin
coating have demonstrated improved patency and target
lesion revascularization rates in the femoral-popliteal seg-
ment, especially when they are appropriately sized and
expanded to the underlying vessel (Fig. 7).
9
Although a
significant correlation between the number of patent runoff
vessels and stent patency is under debate, patients'
underlying tibial disease as well as the concern for occlud-
ing collateral vessels has spurned many away from using
covered stents in popliteal lesions in patients with CLI.
The Supera stent (Abbott Vascular, CA) is a uniquely
woven nitinol stent that has demonstrated significantly
improved radial strength and crush resistance when com-
pared with the traditional nitinol stents. Recent trials in the
popliteal artery have demonstrated superior patency rates to
traditional nitinol stents in the popliteal artery. The rate of
stent fracture with this type of stent has been reported as
extremely low.
10
The data from the randomized Surgical
Versus Percutaneous Bypass Trial are pending publication.
Recognizing and Treating
Complications
Thromboembolic events, restenosis, stent stenosis, and
stent fractures are the main complications of endovascular
Figure 5 A 79-year-old woman with a right foot ulcer (Rutherford category 5). (A) Digital subtraction angiogram (DSA)
demonstrates occlusion of the proximal popliteal artery. (B) The lesion was crossed with the aid of a wire and a catheter, and
debulking was achieved using an orbital atherectomy device. (C) The popliteal artery was then treated with a 6-mm 120-
mm balloon catheter (arrow). (D) Postprocedural DSA demonstrates resolution of stenosis without flow-limiting dissection.
M.R. Rajebi and C. Peña
174
 6
6
 7
7
1
/
7
100%